Polarity is a fundamental concept in chemistry and physics, as it explains how atoms and molecules interact with each other and with external forces. Polarity can affect the physical and chemical properties of substances, such as their solubility, melting point, boiling point, conductivity, and reactivity.
What is polarity and how is it measured?
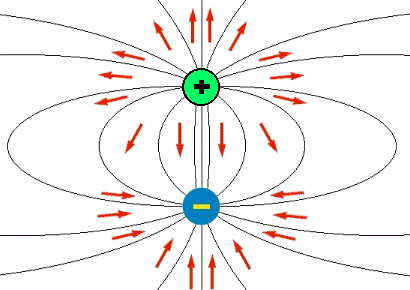
Polarity is the distribution of electric charge among the atoms or molecules that make up a substance. When atoms or molecules share electrons in a chemical bond, they may do so unequally, resulting in a partial positive charge on one end and a partial negative charge on the other end. This creates a dipole, or a pair of opposite charges separated by a distance.
The degree of polarity of a bond or a molecule depends on the difference in electronegativity between the atoms or groups involved. Electronegativity is a measure of how strongly an atom attracts electrons to itself. The more electronegative an atom is, the more it pulls electrons away from other atoms.
The polarity of a bond can be expressed by its dipole moment, which is the product of the charge difference and the distance between the charges. The polarity of a molecule can be expressed by its net dipole moment, which is the sum of all the individual bond dipoles in the molecule.
What are the types and examples of polarity?
Polarity can be classified into two main types: bond polarity and molecular polarity.
Bond polarity refers to the polarity of a single chemical bond between two atoms. A bond can be either polar or nonpolar, depending on the electronegativity difference between the atoms. A polar bond has a significant electronegativity difference, resulting in a partial positive charge on one atom and a partial negative charge on the other atom. A nonpolar bond has a negligible electronegativity difference, resulting in no charge separation.
For example, in a hydrogen-chlorine (H-Cl) bond, chlorine is more electronegative than hydrogen, so it pulls electrons away from hydrogen, creating a polar bond with a partial negative charge on chlorine and a partial positive charge on hydrogen. In contrast, in a hydrogen-hydrogen (H-H) bond, both hydrogen atoms have the same electronegativity, so they share electrons equally, creating a nonpolar bond with no charge separation.
Molecular polarity refers to the polarity of a whole molecule composed of multiple atoms or groups. A molecule can be either polar or nonpolar, depending on its shape and symmetry. A polar molecule has an asymmetrical shape and an uneven distribution of charge, resulting in a net dipole moment. A nonpolar molecule has a symmetrical shape and an even distribution of charge, resulting in no net dipole moment.
For example, water (H2O) is a polar molecule because it has a bent shape and two polar bonds that do not cancel each other out, creating a net dipole moment with a partial negative charge on oxygen and a partial positive charge on each hydrogen. In contrast, carbon dioxide (CO2) is a nonpolar molecule because it has a linear shape and two polar bonds that are opposite and equal to each other, canceling each other out and creating no net dipole moment.
Why is polarity important?
Polarity is important because it determines how substances interact with each other and with external forces. Some of the effects of polarity are:
Chemical polarity
Polarity affects solubility: Solubility is the ability of a substance to dissolve in another substance. Generally, substances with similar polarity tend to dissolve in each other, while substances with different polarity tend to repel each other. This is because polar substances attract each other through intermolecular forces such as hydrogen bonding, dipole-dipole interactions, and ion-dipole interactions, while nonpolar substances attract each other through weaker forces such as London dispersion forces. For example, water is polar and oil is nonpolar, so they do not mix well together.
Chemical polarity
Polarity affects melting point and boiling point: Melting point and boiling point are the temperatures at which a substance changes from solid to liquid and from liquid to gas respectively. Generally, substances with higher polarity tend to have higher melting points and boiling points than substances with lower polarity. This is because polar substances have stronger intermolecular forces that hold them together more tightly than nonpolar substances. For example, water has a higher boiling point than methane because water has stronger hydrogen bonds than methane has London dispersion forces.
Chemical polarity
Polarity affects conductivity
Conductivity is the ability of a substance to allow electric current to flow through it. Generally, substances with higher polarity tend to have lower conductivity than substances with lower polarity. This is because polar substances have more charge separation and more resistance to the movement of electrons than nonpolar substances. For example, water has a lower conductivity than copper because water has more charge separation and more resistance to the movement of electrons than copper.
(India CSR)